U.S. Multicenter Clinical Trial of Corneal Collagen Crosslinking
As lead author of the two studies leading to approval and medical monitor of the U.S. clinical trials of crosslinking , Dr. Peter Hersh and CLEI are gratified that this important new keratoconus treatment will now be more widely available. In December, 2021, Dr. Greenstein and Dr. Hersh published the article, “Corneal Crosslinking for Progressive Keratoconus and Corneal Ectasia: Summary of US Multicenter and Subgroup Clinical Trials.
Click article below to read:

At the CLEI Center for Keratoconus, we started the crosslinking procedure in February 2008 and thank our many patients who participated in the study. If you have any questions, please contact us at 201-692-9434 or email info@www.vision-institute.com. To read the actual clinical study, click on the title above. The video below, summarizes our results.
How Corneal Collagen Crosslinking Works
The goal of corneal collagen crosslinking (CXL) is to decrease the rate of progression of keratoconus, to improve your visual prognosis. In short, corneal crosslinking makes the cornea stronger. Keratoconus is a progressive process in which the cornea (the clear dome-shaped front of the eye, like a watch crystal) deforms in association with thinning and biomechanical weakening.
This is much like a bulge that can form on a thin area of a car tire or balloon. This progressive distortion and bowing out (coning) of the normal cornea creates optical aberrations, which can be thought of as static in your eye (like static on an old TV), resulting in a decrease in visual function. Because of the corneal distortion in keratoconus, patients usually require rigid or complex curvature contact lenses to achieve good functional vision; spectacle correction frequently does not result in an acceptable quality of vision.
Given the distortion of the cornea, contact lens fitting may be challenging in many patients. Also, since keratoconus tends to progress over the second to fifth decades of life, it can lead to intolerance of contact lenses and, ultimately, necessitate corneal transplantation. So, a treatment to slow or stop keratoconic progression would be very important to the keratoconus patient.
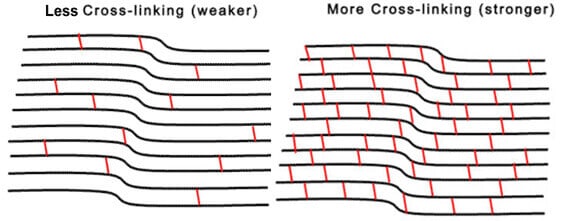
Since we began treating patients with crosslinking nearly a decade ago in our clinical studies, CXL has emerged as an important advancement in keratoconus treatment to slow or stop the progression of keratoconus. In this procedure, riboflavin (Vitamin B2) is administered in conjunction with ultraviolet A (UVA – 365nm) light.
The interaction of riboflavin and UV causes a photobiologic reaction in the corneal leading to the formation of additional molecular bonds between collagen molecules as well as their supporting biologic sugars. This results in stiffening of the cornea, much a placing additional cables on a bridge will help to support it. In the video below, Dr. Hersh discusses the principles of corneal cross-linking.
The Corneal Collagen Crosslinking Procedure
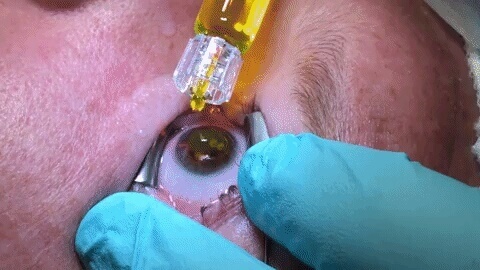
Corneal collagen cross-linking involves the following steps:
- After numbing drops are applied, your eyelids will be held open with a small instrument to avoid blinking during the procedure.
- The outer layer of cells of the cornea (clear covering of the eye) will be removed by either a manual or laser technique. Sometimes we will leave this layer in place (the epi-on procedure), depending on your individual situation.
- The lid holder is removed and riboflavin (vitamin B2 eyedrops) will be applied to your eye until there is full uptake into the cornea.
- The ultraviolet (UV-A) light source is then aligned and then you will look up at it for administration of the crosslinking treatment.
- At the end of procedure, a bandage soft contact lens will be placed on your eye, to protect the surface of the cornea during the healing process.
What to Expect after Crosslinking
As discussed by Dr. Hersh in the video above, the first phase of healing is the early period 1-5 days when the surface epithelial cells heal. Generally, a bandage contact lens will protect the cornea and make it comfortable during this time.
However, there may be some discomfort for which we will give you the appropriate medication. You will be able to see through the lens, but vision will be hazy during this time (like driving with a foggy windshield). In some patients, vision may continue to be mildly hazy for several months, but, in general, most patients note a return to their baseline vision over the first few weeks.
Contact lenses can be restarted after we are satisfied with the early healing, usually around 1 month after the procedure. Remember, collagen crosslinking is designed to decrease the progression of keratoconus and corneal ectasia. It is not meant to improve your vision.
There are other procedures that can be used to further improve your vision. It is important to know that, although crosslinking has been shown to be successful in most patients, it may not work in all cases and keratoconus may still continue to progress.
Crosslinking Results
Corneal collagen crosslinking is a very successful procedure, with over a 90% success rate of decreasing the progression of keratoconus. Here at CLEI, we have been working on crosslinking for 10 years. The results of the U.S. clinical trial of corneal collagen crosslinking are discussed by Dr. Hersh in the video below.
The following abstract is taken from the clinical study paper of crosslinking written by Dr. Hersh.
Purpose: To evaluate the safety and efficacy of corneal collagen crosslinking (CXL) for the treatment of progressive keratoconus.
Design: Prospective, randomized, multicenter, controlled clinical trial. Participants: Patients with progressive keratoconus(n=205).
Methods: The treatment group underwent standard CXL and the sham control group received riboflavin alone without removal of the epithelium.
Main Outcome Measures: The primary efficacy criterion was the change over 1 year of topography-derived maximum keratometry value (ie the topographic height of the keratoconic cone), comparing treatment with control group. Secondary outcomes evaluated were corrected distance visual acuity (CDVA), uncorrected distance visual acuity (UDVA), manifest refraction spherical equivalent, endothelial cell count, and adverse events.
Results: In the CXL treatment group, the maximum keratometry value decreased (improved), on average, by 1.6 diopters (D) from baseline to 1 year, whereas keratoconus continued to progress in the control group. In the treatment group, the maximum keratometry value decreased by 2.0 D or more in 28 eyes (31.4%) and increased by 2.0 D or more in 5 eyes (5.6%). The CDVA improved by an average of a little over one line on the vision chart. Twenty-three eyes (27.7%) gained and 5 eyes lost (6.0%) 2 lines or more. The UDVA improved by nearly one line on the vision chart. Corneal haze was the most frequently reported CXL-related adverse finding. There were no significant changes in endothelial cell count 1 year after treatment.
Conclusions: Corneal collagen crosslinking was effective in improving the maximum keratometry value, CDVA, and UCVA in eyes with progressive keratoconus 1 year after treatment, with an excellent safety profile. Corneal collagen crosslinking affords the keratoconic patient an important new option to decrease the progression of this ectatic corneal process.
Who Should Get Crosslinking?
Crosslinking is designed specifically to decrease the progression of keratoconus, to prevent worsening of vision. Some patients tend to respond better while others don’t require treatment. We have done a study to determine who are the best candidates as described in the video below.
For a discussion of the epi-on crosslinking procedure, watch the presentation below.
Discussion of the crosslinking clinical trial written by Dr. Hersh:
Crosslinking is thought to biomechanically strengthen the corneal stroma and, consequently, slow the progression of corneal ectatic disease processes. In the corneal crosslinking procedure, the photobiologic process depends on the interaction of riboflavin with ultraviolet A (UVA – 365 nm) irradiation, with the formation of reactive species that interact with corneal proteins and lead to the formation of chemical bonds.7-11 In both in vitro and clinical studies of the cornea after CXL, a number of morphologic and physiologic changes have been reported.12-17 Indeed, studies show that, immediately after crosslinking, stress measurement increases in human corneas by over 300%.18
By means of this corneal biomechanical strengthening, the essential clinical goal of corneal collagen crosslinking in keratoconus is to decrease disease progression over time. Since the severity of keratoconus is generally related to the degree of steepening and irregularity of the corneal optical architecture, the primary efficacy outcome for this study was based on maximum K, as measured by a Scheimpflug corneal topography system. Though not measuring all aspects of keratoconus severity, the topographic maximum K reflects the severity of the keratoconic cone, and affords an objective, quantitative endpoint for analysis that can be standardized amongst multiple study sites.
The change in maximum K over time, therefore, serves as a quantitative indicator of a change in keratoconus severity. A difference of at least 1.0 diopter in the mean change in maximum K from baseline to one year, comparing the treatment and control group, was chosen as a clinically meaningful endpoint of study success. Thus, either stabilization or improvement in maximum K would indicate a positive effect of the procedure on disease evolution, whereas an increase in maximum K would suggest continued progression.
Topography Changes after Crosslinking
The mean maximum K value of the crosslinking treatment group decreased by 1.6 D at 1 year, compared with 1.0 D steepening of the control group, a difference of 2.6 D between treatment and control. Thus, our study demonstrates that crosslinking has a beneficial effect on corneal topography in keratoconus patients over one year. This salient finding is in agreement with a number of international clinical trials using a similar protocol, which found decreases in maximum K value of 2.01 D3 1.90 D,19 1.46 D,20 and 1.42 D21 after crosslinking. In addition to improved maximum K, other topography and wavefront analyses of a single center cohort of patients by one of the authors (PSH) demonstrate a more general optical improvement after CXL, finding that quantitative descriptors of corneal topography22, as well as both corneal and total ocular aberrations23, improve one year after crosslinking.
In addition to the standard metric of mean change in maximum keratometry, it is, clinically, perhaps clearer to the patient if we present results as proportions of patients with different outcomes; this may elucidate the patient’s understanding and better define procedure expectations. It is, therefore, helpful to look at the likelihood that an individual patient will improve or decline substantially. In our study, the maximum K value decreased by 2.0 D or more in 28 eyes (32%) and increased by 2.0 D or more in 5 eyes (6%) one year after crosslinking. Although, these latter 5 eyes might be considered treatment failures because cone progression was not stabilized24, it is unclear what the natural evolution of the disease might have otherwise manifested; it is possible that disease progression was slowed but not completely, or that progression, indeed, proceeded apace. In an effort to define preoperative characteristics which might influence outcomes, additional reports from one author (PSH) suggest that eyes with preoperatively steeper corneas25, as well as corneas with more central cones26, have a greater likelihood of topography improvement 1 year postoperatively. The only independent predictor of improvement in maximum K after crosslinking was preoperative maximum K; eyes with a maximum K >55D were 5.4X more likely to have topographic flattening >2D after CXL compared to eyes with flatter corneas; there were no independent predictors of failure of the procedure to stabilize the cornea, however.25
Vision Changes after Crosslinking
In addition to the primary efficacy measurement of maximum K, changes in CDVA may point to additional benefits or, conversely, to safety concerns after crosslinking. Since decrease in vision in keratoconus results from a distorted corneal optical architecture, topography improvement would be expected to yield improved visual function. Indeed, in this study, crosslinking was associated with an improvement of more than 1 line of mean CDVA 1 year postoperatively. Although this finding was statistically significant, the actual clinical significance is better demonstrated in studying the outcomes on an individual basis; among patients receiving CXL, twenty-three eyes (24%) gained 2 or more lines of CDVA, whereas 5 eyes (6%) lost 2 lines or more. Thus, nearly one quarter of patients enjoyed a clinically meaningful increase in CDVA as a result of crosslinking, whereas some eyes do continue to progress. Our findings are in agreement with previous studies by Vinciguerra and associates27, which found that mean CDVA improved between one and two logMAR lines at 12 months postoperatively. Similarly, at 1-year follow-up, Caporossi’s group28 and Raiskup-Wolf and colleagues20 found improvements in CDVA, noting continued improvement even after 1 year.
With regard to those 5 eyes in our study who lost 2 or mores lines CDVA, 2 showed continued topography progression, 2 had residual corneal haze or scar, and one had no potentially contributing cause. In the aforementioned multifactorial analysis25, the only independent predictor of a change in postoperative CDVA after CXL was the preoperative corrected distance visual acuity; those eyes with worse preoperative CDVA (<20/40) were 5.9X more likely to experience an improvement in vision one year after crosslinking. However, with regard to eyes which lost vision from the procedure, the most salient indicator of an unwanted outcome, there were no independent preoperative predictors.
Analysis of UDVA showed slightly less than 1 line average improvement in our study. This improvement is somewhat less than previously reported. Vinciguerra’s group27 reported a significant improvement in mean UDVA, from 0.77 logMAR preoperatively to 0.57 logMAR 1 year postoperatively. Similarly, Caporossi and colleagues29 reported a significant improvement in mean UDVA of 2.41 Snellen lines. In addition to these visual acuity improvements, it is notable that many patients appreciated a subjective improvement in a number of measures of their visual function after the crosslinking procedure.30 (Figure 6) Although we did find statistical significance in these subjective improvements, the actual clinical impact of these improvements remains unclear.
Clinical Time Course after Crosslinking
It is important, clinically, for the ophthalmologist to understand the tempo of healing and outcomes evolution after crosslinking, both to identify untoward effects and to properly counsel the patient’s expectations. Topography and vision outcomes seem to follow a reproducible time course after treatment.31 Looking at the changes over time in the crosslinking treatment group, there was a worsening in maximum K at 1 month, with improvement thereafter, continuing over one year (Figure 3). The time course of CDVA change was similar, with a slight drop at one month, followed by improvement (Figure 4).
This clinical time course after CXL reflects that of the natural history of crosslinking associated stromal haze (discussed below) and corneal thickness measurements36, suggesting that wound healing responses continue over months concomitant with the evolution in clinical outcomes. Both epithelial and stromal healing and remodeling mechanisms may impact this time course of outcomes. The epithelium tends to mask the stromal cone of keratoconus. 32,33 With corneal de-epithlialization in the CXL procedure, the more profound stromal irregularity is revealed, with an increase in measured maximum K. As the epithelium heals and remodels over months, topography improvement is seen. Stromal healing, too, may play a part in the post-CXL timecourse. For instance, both in vitro and ex vivo studies have shown that collagen crosslinking leads to an almost immediate loss of keratocytes in the corneal stroma, followed by repopulation over the next six months.13,34,35
Safety of Corneal Crosslinking
Adverse event analyses in this study suggested an excellent safety profile for the crosslinking procedure. Corneal stromal haze was the most frequently reported adverse event in the study. Indeed, haze appears to be a normal concomitant of the crosslinking procedure. Typically, haze is first observed as dust-like change in the anterior corneal stroma which evolves into a mid-stromal demarcation line.37 Crosslinking associated corneal haze is most likely a result of back-scattered light, causing decreased corneal transparency, and likely demarcates the depth of the actual crosslinking effect.38 Previous analysis a single center cohort by one author (PSH) found that crosslinking associated haze peaks at 1 month, plateaus between 1 and 3 months, clears between three and six months, and continues to return to baseline at one year; thus, this reflects the clinical time course of CXL outcomes .36,39 Corroborating these results, in the current study, only 3 eyes remained with stromal haze at one year postoperatively. To date, it remains unclear whether postoperative haze is an unwanted complication or rather a desired wound healing effect demonstrating, and perhaps enhancing, the efficacy of the crosslinking procedure. Although there were 2 eyes in our study with a decrease of CDVA in the presence of ongoing haze, previous work has not shown a correlation between corneal haze with either topography or visual acuity outcomes.39
Endothelial cell damage from crosslinking is a concern, and could potentially result from endothelial exposure to free radicals generated from the crosslinking process. The original parameters for CXL suggested a minimum stromal thickness of 400 um to attenuate the UVA power and thus prevent endothelial damage.40,41 Indeed, in the study protocol, corneas needed to be swollen with a hypotonic riboflavin solution to the 400 um threshold before proceeding with UV exposure.42,43 Results herein showed no damage to the corneal endothelium and no reports of corneal decompensation after crosslinking.
Microbial keratitis likely is one of the most severe potential complications of CXL. In this study, there was one reported corneal ulcer which occurred in a patient initially assigned to the control group, who subsequently had the CXL procedure. Since a total of 293 eyes ultimately received crosslinking in this trial, the incidence was 0.3%. There have been several reports of microbial keratitis after CXL in the literature, including bacterial, fungal, acanthamoeba, and herpetic.44 Given the epithelial defect and use of a therapeutic contact lens in the crosslinking procedure, care should be taken to assure rapid re-epithelialization, treat ocular surface pathologies, and maintain careful postoperative followup to detect early complications.45
Conclusions
Concluding, this randomized, controlled clinical trial demonstrates the efficacy and safety of corneal collagen crosslinking for the treatment of progressive keratoconus. In addition to decreasing disease progression, crosslinking also can have beneficial visual and optical effects such as decrease in corneal steepness and improvement in visual acuity and subjective visual function in some patients. Although this trial assesses results over a one year period, international studies have reported good stability over 10 years.46 Indeed, the beneficial clinical effect of crosslinking is highlighted by Sanvik and colleagues47, who report a more than a 50% decrease in the frequency of keratoplasty in keratoconus patients in Norway, comparing a period before crosslinking was available with a similar period after crosslinking became well established. Certainly, the topography and visual results reported herein support the efficacy of crosslinking in the stabilization of this progressive corneal disorder.